The reaction between 1,2-epoxy-1-methylcyclohexane with hydrogen bromide yields 2-bromo-2-methylcyclohexanol as the major product. Justify with appropriate mechanistic route. | Homework.Study.com
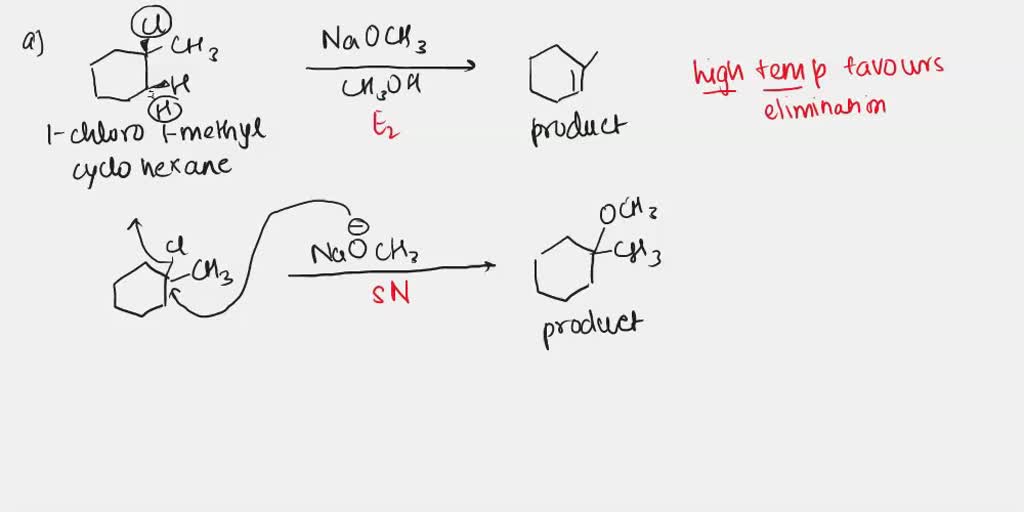
SOLVED: a) 1-chloro-1-methylcyclohexane reacts with NaOCH3 in methanol to form a product mixture. Both the product of elimination and the product of nucleophilic substitution are formed. Include the mechanisms and products of

Draw the products of the reaction of 1-methylcyclohexene with each of the following reagents. Be sure to indicate stereochemistry where necessary or pertinent. A) HCl B) HBr C) HI D) HCl/H_2O_2 E)

Name of the alkene which will yield/chloro 1-methylcyclohexane by its reaction with HCl. Write the - YouTube
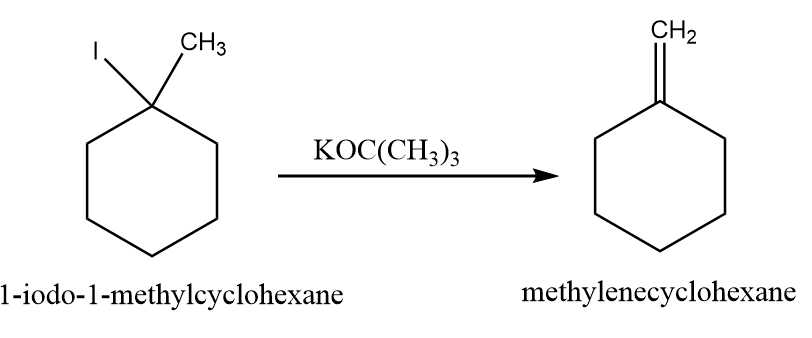
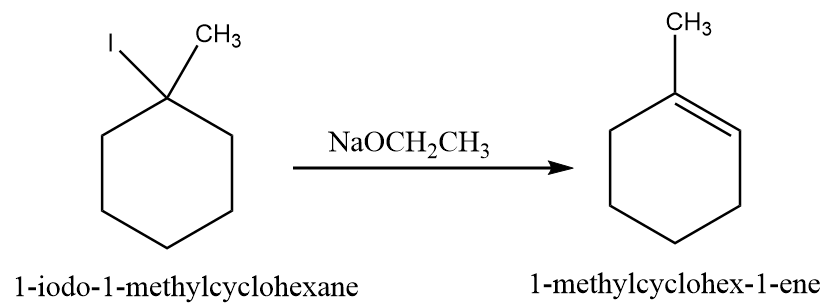
When 1-iodo-1-methylcyclohexane is treated with $ NaOC{H_2}C{H_3} $ as the base, the more highly substituted alkene product predominates. When $ KOC{(C{H_3})_3} $ is used as the base, then less substituted alkene predominates.

Assume that you are carrying out the dehydration of 1-methylcyclohexanol to yield 1-methylcyclohexene. How could you use infrared spectroscopy to determine when the reaction is complete? | Homework.Study.com